Analysis of works of art has always been complex due to their often delicate nature and their cultural and historical relevance. However, such analysis is necessary to investigate their constituent materials and, possibly, their authenticity. Some compounds serve as chronological indicators of an artwork’s production. Identifying materials typical of the 20th century (such as titanium white or cadmium red) or of the 19th century (such as zinc white, cobalt blue, cadmium yellow, or chromium-based pigments) can therefore provide a terminus post quem for the analyzed piece. The existing scientific literature includes a wide range of studies on works of art devoted to the identification of forgeries and restorations1,2, particularly in paintings3, but also on other materials such as metals3, ceramics4, enamels5, and manuscripts6.
For this purpose, non-destructive and non-invasive analytical methods are crucial, as they make it possible to obtain valuable information without damaging the artworks or taking samples. Among these, portable X-ray fluorescence (pXRF) has recently gained a prominent role thanks to its versatility, speed, and ability to perform in situ analysis7.
The XRF technique: principles, variants, and evolution
X-ray fluorescence spectroscopy (XRF) is a well-established technique for elemental analysis. It operates by exciting atoms in a sample with a primary X-ray beam. The penetrating power of X-rays enables the analysis to probe beyond the surface, providing information from the sub-surface layers (bulk analysis)8. As excited atoms return to their ground state, they emit characteristic X-rays. The wavelength identifies the element, while the intensity is proportional to its concentration. It is crucial to note that this intensity is influenced by factors such as the element’s fluorescence yield, competing non-radiative decay processes, and matrix absorption effects8,9,10.
This technique and numerous specific variants are referred to in various ways such as X-ray Fluorescence, XRF Spectrophotometry, EDXRF (energy dispersive XRF, it is one of the most common detectors used, the “ED” is often omitted)11, µ-XRF (micro-XRF, utilizing capillary lenses or collimators to achieve micrometric spatial resolution)12, MA-XRF (macro XRF, large-scale XRF scanning)10, AR-XRF13 (angle-resolved XRF, which uses varying incident/detection angles to probe different depths), etc. The portable version of the instrument is sometimes referred to as pXRF1, even though the concept of portability is difficult to define (e.g., from what weight onwards is an instrument considered portable, or when it comes to a portable instrument, rather than just transportable?). Portable XRF is therefore not generally considered a true technique in its own right. However, the operating principle is that of the XRF technique. The most recent shift has been the rise of HandHeld XRF (HH-XRF) over the past 15–20 years, i.e., a very compact portable XRF that can be held directly by hand. These all-in-one devices maximize portability and operational speed, though often at the cost of spectral resolution and light-element sensitivity compared to bulkier portable systems13. The frontier of the technique now includes the use of Artificial Intelligence (AI) for data interpretation and mining14. The effective application of AI, however, critically depends on robust training with curated datasets to prevent analytical errors and ensure reliable outcomes13,15.
A critical review of the evolution of XRF systems is necessary, as each successive technology has brought distinct advantages and constraints. This historical perspective is vital to contextualize and interrogate the vast body of legacy data encountered in the literature. Moreover, this same rigorous, critical framework must be applied to contemporary data, ensuring that the limitations of modern systems are also acknowledged and addressed.
The use of a primary X-ray beam to induce fluorescent radiation in a sample was first proposed by Glocker and Schreiber in 192816. In the 1950s and 1960s, the XRF technique was pioneered in the field of cultural heritage, especially for the study of metal alloys17 and at least from the 1970s for the study of pigments18,19 and many other materials of historical and artistic interest17,20. In the 1980s, the XRF technique began to be listed in the equipment of museums17. In the following years, safer instruments progressively became available, with increasingly smaller dimensions and lower costs, which also present notable operational simplifications, especially in terms of X-ray production systems, types of detectors, and cooling2. Already in 1996, instruments equipped with portable X-ray tubes were described (therefore without the need for radioactive sources) and with a Silicon PIN diode cooled thanks to the Peltier effect (therefore no longer needing refrigerants such as liquid nitrogen), with a great improvement in terms of safety and dimensions, and with a weight of around just 8 kg11,20. With the new millennium, the serial production of truly portable commercial instruments began. These developed from a wide range of early prototypes built by Universities and Research Institutions21,22,23, which were often more “transportable” than portable. Subsequently, XRF tools have gradually become practically indispensable in any laboratory of conservation science20. It should not be overlooked that the achievement of portability initially led to a decrease in performance, especially in terms of the dimensions of the spot of analysis and the ability to identify light elements20. Until a few years ago, the lower limit was generally Z = 15 (phosphorus)17,24, while in recent years the range of identifiable elements has expanded. Many of the instruments currently on the market are able to detect the presence, in a semi-quantitative way, of elements with atomic numbers starting from 12 (magnesium)25. Using the helium flow device, it is also possible to identify sodium (Z = 11) and generally improve the obtainable signal24.
Regulatory framework and professional standards
It is also crucial to acknowledge that, despite significant technical advancements, XRF instrumentation implies the use of ionizing radiation, making strict adherence to radiation safety protocols mandatory. While modern portable systems are undoubtedly safer than their 20th-century predecessors (primarily due to the move away from radioactive sources), the regulatory landscape governing the use and transport of X-ray generating equipment remains stringent and varies by jurisdiction26. These regulations have, at times, constrained the adoption of the latest technologies. A pertinent example is that legal limits on X-ray tube energy in some European countries long restricted systems to 25 keV, preventing the use of more powerful 50 keV tubes. This technical limitation directly impacts analytical performance, resulting in a lower penetration depth and a reduced ability to excite heavier elements. Consequently, data acquired with these restricted systems is inherently less comprehensive, a critical factor that must be accounted for when interpreting or utilizing such legacy data.
Furthermore, the execution of XRF analysis and the interpretation of its data are subject to professional standards. As a physicochemical technique, it requires specific expertise. Italian law, for example, stipulates that expert reports and assignments in pure and applied chemistry for Judicial Authorities and Public Administrations can only be entrusted to individuals registered with the National Federation of Chemists and Physicists, or to those otherwise qualified to perform such analyses27,28. This is particularly appropriate for analyses with legal implications, such as authentication, attribution, or the identification of forgeries, where the analyst may be called upon to answer to a Judicial Authority.
This underscores a fundamental synergy: the professional’s expertise is indispensable for the correct interpretation of complex data, just as the researcher’s scientific insight is vital for driving technological innovation. Therefore, when evaluating historical data, it is critical to consider the technological and regulatory context in which it was generated, as well as the competencies of those who produced it. Looking forward, a truly multidisciplinary approach, bridging the gap between research-driven development and professional application, is essential to ensure that the data we produce today will remain a robust and meaningful resource for future generations.
Inherent limitations of XRF for heritage applications
Despite the several advantages XRF analysis brings to this scope, as highlighted by recent comprehensive reviews on XRF use (Monico et al.8, Orsilli et al.13, Potts et al.15), the interpretation of data obtained can be difficult and ambiguous. The technique has many limitations that can reduce its effectiveness, and these limitations are often overlooked.
For a true quantitative analysis, it would be necessary to operate with appropriate reference standards of known chemical composition, precise and closely similar to that analyzed17. Such standards are difficult to obtain or simply not available (for example, consider all the possible mixtures of pigments). In fact, even in those cases (such as metal alloys) where it is certainly easier to find them, such standards are rarely used. The challenge of quantification extends beyond the availability of standards. Even when appropriate references exist, two fundamental obstacles complicate accurate concentration determination: matrix effects and unknown layer geometry. Matrix effects arise from differential X-ray absorption by different materials: the same elemental concentration produces vastly different signal intensities depending on the surrounding matrix. For instance, identical iron content in lead white (oil painting) versus calcium carbonate (fresco) yields dramatically different peak intensities. The unknown thickness of stratified layers compounds this issue, making the exact analyzed depth indeterminate. For these reasons, semi-quantitative analysis often represents the practical approach in heritage studies. By comparing relative peak intensities and elemental ratios across multiple measurement points, meaningful diagnostic patterns emerge without requiring absolute concentrations, which is generally sufficient for pigment identification and authentication purposes.
With the results obtained with XRF alone, however, we can only hypothesize the nature of the materials. All this because XRF is an elementary technique and, unlike other analytical methodologies, it is not able to establish how the elements it identifies are linked to each other2. For this reason, the identification of pigments often relies on a deductive method, supported by contextual information such as the overall chromatic appearance of the analyzed area17. A red containing mercury is most likely composed of cinnabar or vermilion, but it cannot be ruled out, for example, that other pigments, such as red lacquers or Hansa reds, may also be present (perhaps as part of a restoration or forgery!). In this case, nothing could be done but to formulate credible hypotheses, without ever forgetting that these are still results of interpretation and therefore with a certain degree of subjectivity. All of this is a consequence of another major limitation of portable XRF technology: the inability to identify individual light elements. Light elements can only be estimated overall, but they could constitute any colorant, perhaps adsorbed onto other inorganic compounds. Organic compounds completely or almost completely elude XRF analysis and are capable of radically modifying the overall chromatic appearance, potentially undermining the entire deductive process.
The multi-analytical framework
Crucially, especially on handheld instruments, specific calibration programs for quantitative and even qualitative calculations based on standard matrices (e.g., precious metals or geological samples) are often employed; however, their direct application to complex, stratified matrices like paintings in Cultural Heritage is inherently imprecise and inevitably introduces a significant degree of error15. For all these reasons, using other analytical techniques in conjunction with XRF, such as Raman spectroscopy (thanks to its high spatial resolution for analyzing minute areas and individual layers, and for unambiguously distinguishing compounds and phases via their characteristic spectra)29. FORS (which identifies pigments by their characteristic spectra, similar to Raman spectroscopy but with lower spatial resolution, and can be performed in situ), SEM-EDS (which can detect elements not clearly identifiable by pXRF and provides precise layer analysis via high-magnification imaging), LA–ICP–MS (which has a low spatial resolution and allow isotopic analysis and multielement quantification)30 or techniques such as hyperspectral imaging is often essential to avoid making serious mistakes in interpretation.
For all these reasons, the integration of pXRF with complementary techniques is not merely beneficial but methodologically necessary: treating pXRF as a standalone technique risks significant misinterpretation. Looking forward, advanced modalities such as MA-XRF, AR-XRF, and the integration of artificial intelligence (AI) for data interpretation hold the promise of overcoming current limitations. However, to effectively train these AI models and prevent the perpetuation of misinterpretations, it is crucial to ground them in reliable data. This requires a critical re-evaluation of the existing XRF literature to build robust interpretive frameworks for the future.
Given these constraints, this review focuses specifically on pigments in paintings and, briefly, on metal alloys, which are material classes where XRF demonstrates diagnostic value since they typically contain heavy elements readily detected by XRF. Materials such as ceramics and glass, though analyzable by XRF, present additional complexities, such as predominance of light elements, more ambiguous chronological markers, and complex firing/manufacturing histories, that would require extensive separate treatment beyond the scope of this practical guide.
Case studies
The theoretical limitations discussed (stratigraphic ambiguity, the elemental nature of the data, matrix effects) find clear manifestation in the practical analysis of real artifacts. The following case studies, drawn from the authors’ experience, serve to concretize these concepts and demonstrate why a purely elemental approach, based on pXRF alone, is often insufficient for reaching robust diagnostic conclusions.
Case one – Sarcophagus of Ramose
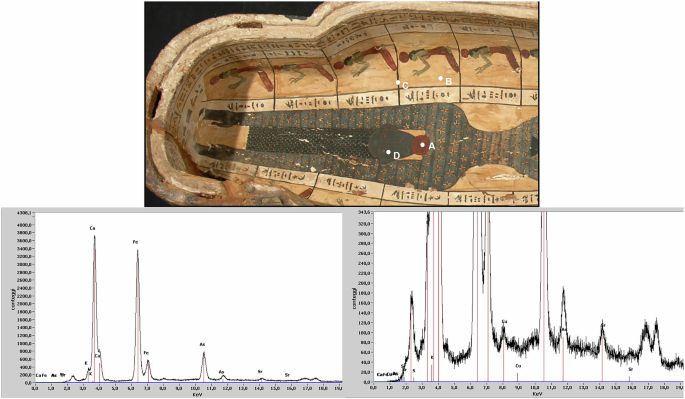
A non-invasive analysis using portable X-ray fluorescence (pXRF) spectroscopy was performed on the sarcophagus of Ramose (XXVI Dynasty), housed in the Museum of the City of Narni (Umbria, Italy), to identify and compare the pigments and materials used. The spectrum shown in Fig. 1, for instance, is representative of measurement point A31. The analysis detected calcium (Ca), iron (Fe), and arsenic (As) as the main elements, along with minor peaks of sulfur (S), potassium (K), strontium (Sr), and copper (Cu).

Analyses performed using an Assing LITHOS 3000 portable spectrometer. The instrument was equipped with a Mo-Kα excitation source, operated at 25 kV and 0.3 mA, and a Peltier-cooled Si-PIN detector.
While a straightforward reading of the signals might suggest a painted layer of red ocher (Fe) and realgar (As, S) applied over a gypsum-based preparation (Ca, S), this remains highly speculative. A single-point analysis presents too many unknowns for a reliable interpretation, especially if the instrument cannot stratigraphically distinguish overlapping layers or establish the depth at which an element is located, making it impossible to definitively assign specific elements to either the preparation layer or the paint layer itself. To improve the reliability of the data, one can perform comparative analyses at multiple points on the artwork, evaluating the consistency of peak intensities and the ratios between specific elements. However, as the following case will demonstrate, it is often not possible to reach a definitive and unambiguous conclusion. The analytical process, therefore, frequently relies on plausible reconstructions and hypotheses, which are always subject to revision. In this specific case, for instance, arsenic (As) was consistently detected at all four points marked in Fig. 1, as well as at other unmarked locations. This recurring presence supports a reasonable hypothesis that the arsenic is part of the preparation layer. This hypothesis seems to be confirmed by the results from areas where the underlying ground is directly accessible for analysis, such as point B in Fig. 1. However, alternative interpretations must be considered since ubiquitous arsenic could also be a residue from a 19th-century conservation treatment, as arsenic compounds were commonly used as wood preservatives31,32. The case of iron is even more complex. While the preparation layer likely contains an earth pigment based on iron oxides/hydroxides (or even sulfates)31,33, attributing the yellow color specifically to yellow ocher would be premature. This color could equally stem from organic compounds (such as watercolors or patinating agents applied during a restoration) or even from arsenic-based pigments. Once again, the stratigraphic origin of these elements remains uncertain, and we cannot rule out the presence of multiple, compositionally distinct preparation layers that may also contain Fe and As. An additional complicating factor is potential contamination: iron is a common component of excavation soils and dust, which can deposit in surface cracks and be detected by XRF at virtually any point of analysis. Ultimately, distinguishing conclusively between these numerous possibilities using XRF data alone is often technically unfeasible. Nevertheless, many authors have drawn, and continue to draw, definitive conclusions from single XRF analyses, even though the limitations of such an approach are widely acknowledged.
Further analysis of Fig. 1 reveals additional critical issues. The black line of the score (point C) contains Fe. However, this does not imply the presence of the rare black iron oxide (magnetite). A more plausible explanation is that the line is composed of a carbon-based black, where Fe is a common impurity and the primary element detected by XRF because of its atomic weight. Alternatively, the Fe signal could originate from the background or result from the analytical spot size exceeding the width of the thin line. This interpretation is supported by the inherent limitations of pXRF in spatial resolution, which is often insufficient to isolate such fine details. The complexity of element interpretation extends beyond carbon-based blacks to historically significant pigments that XRF alone cannot distinguish. In fact, regarding the blue pigment at point D, the presence of Cu is consistent with historical palettes but cannot be used to identify the specific pigment, as most blue and green pigments from this period are copper-based17. Even looking at the presence of other elements, such as silicon and calcium (both often ubiquitous in these contexts), would make it impossible to distinguish between pigments such as azurite and Egyptian blue. This analytical ambiguity is a well-documented challenge, as proved by some representative cases such as the fresco cycle of Castelseprio. A portable XRF campaign in 2009 suggested the use of azurite34, a hypothesis later refuted through complementary techniques like fiber optics reflectance spectroscopy (FORS) and visible induced luminescence (VIL), which identified one of the most significant and rare uses of Egyptian blue instead35. Similar considerations apply to the overlooked use of Egyptian blue in Renaissance paintings, including works by prominent masters such as Raphael36,37 and Perugino38. Paradoxically, while its initial misidentification as azurite demonstrates the limitations of pXRF, the very presence of Egyptian blue in a Renaissance painting is now recognized as a powerful indicator of authenticity. This systematic misinterpretation stemmed from the long-standing assumption that Egyptian blue had fallen out of use after the classical period, a historical bias that not only skewed analytical expectations but also ensured the pigment’s absence from a forger’s palette, as they were both unaware of its historical application and unable to source it before its commercial revival in the late 19th century39. This case underscores that the possibilities are often numerous, and the temptation to draw definitive conclusions from a single XRF analysis must be avoided. This does not mean, however, that we should neglect the potential of the instrument, which is sometimes truly surprising. A key feature of XRF is the very high sensitivity of the technique for some elements, which can be leveraged for more subtle archaeometric investigation if combined with appropriate analytical strategies40. For example, working with specific ratios between the intensities of key element signals can reveal interesting correlations. For instance, the Sr/Ca ratio shown in the spectrum in Fig. 1 could provide specific information on the type of gypsum or calcium carbonate present. However, due to the inherent limitations of pXRF, even this refined approach often necessitates a multi-analytical methodology. Leveraging the synergy with more sensitive techniques is therefore crucial for validation and to obtain more definitive results.
Case two – Oil painting on glass
A primary application of portable XRF (pXRF) in heritage science is pigment analysis17. While it is commonly used to reconstruct an artist’s palette, a particularly decisive use is the so-called ‘pigment test’ for authenticity studies. The detection of modern pigments incompatible with the artwork’s purported period can readily expose a forgery41. Among pigments, white ones are often the most diagnostic for this purpose. This preference stems from several factors, notably the well-documented history of their modern development. For instance, due to its toxicity, lead white was progressively replaced throughout the 19th and 20th centuries by zinc white, barium white, lithopone, and finally titanium whites. The exact dates when the succession of pigments took place are often a source of controversy among scholars. To bring an example, according to Pigment Compendium42, the first commercial artists’ white pigment formulation (titanium white) in Europe appeared in 1919, with only limited experimental availability before 191642. Although a titanium dioxide-based pigment was created as early as 1870 (according to The Grove Encyclopedia of Materials and Techniques in Art43), it was developed for industrial purposes like ship paint, not for fine arts. Without going into further detail, all sources agree that titanium white could not have been present in an 18th-century painting. Therefore, the systematic application of pXRF can be a powerful tool for uncovering a significant number of forgeries.
The oil painting on glass shown in Fig. 2 exemplifies this application. The work, stylistically attributed to the 18th century, was analyzed by portable XRF on behalf of the Carabinieri TPC Turin investigation (2009)—Commander Guido Barbieri. The resulting spectrum reveals Ti as the most abundant element, alongside significant amounts of Zn (likely from zinc white), Ca, and Fe. Crucially, there is no spectral ambiguity with secondary lines of elements associated with white pigments such as barium sulfate and lithopone (Ba). Most significantly, the absence of Pb rules out the use of lead white, which would be expected in an authentic 18th-century painting of this kind. Repeated analyses across multiple white areas yielded entirely analogous results. In this clear-cut case, the presence of titanium white is unequivocal evidence of the painting’s inauthenticity. However, such a definitive conclusion requires caution, as the detection of Ti is not always a reliable indicator of a forgery. A significant co-occurrence of Ti and Pb could suggest extensive repainting during a restoration. Conversely, trace amounts of Ti could originate from natural impurities in earth pigments or clays42 and in kaolin29 or from residual restoration materials trapped in the paint’s cracks and porosity (as in the case of analyses carried out on areas cleaned from previous repainting, which are no longer visible). In these ambiguous scenarios, only specific in-depth analyses (e.g., SEM-EDS on a cross-section) can provide a reliable answer. These interpretive challenges are amplified when analyzing colored pigments, where mixtures are common and can be misleading. Therefore, while the white pigment test offers a powerful and simplified approach, a comprehensive examination of the entire palette is always recommended. This case perfectly illustrates the dual nature of pXRF in authentication: it can provide definitive evidence of forgery in clear-cut anachronisms (like Ti in an 18th-century context), yet it also demands caution, as ambiguous traces of the same element require integration with techniques like SEM-EDS on cross-sections to discriminate between forgery, restoration, or natural impurity.

Carabinieri TPC Turin investigation, 2009. Analyses performed using an Assing LITHOS 3000 portable spectrometer. The instrument was equipped with a Mo-Kα excitation source, operated at 25 kV and 0.3 mA, and a Peltier-cooled Si-PIN detector.
Case three – Transit of Mary
In Fig. 3, an analysis of multiple pigments can be observed, which summarizes the results from a study on the painting “Transit of Mary” (LOVAL collection), attributed to Giovanni Maria Morandi (1622–1717).

Analyses were carried out with a SPECTRO X-SORT handheld XRF spectrometer. This system features a Rh-Kα source operated at 45 kV and a Peltier-cooled Silicon Drift Detector (SDD).
The analysis focused on blue pigments, as distinguishing between Prussian blue, smalt, and ultramarine is crucial for the work’s attribution, dating, and historical contextualization. Initial XRF analyses provided limited clarity; while they detected cobalt in some areas, they could not differentiate between smalt and modern pigments like Thenard’s blue. The combination of pXRF with multispectral imaging (e.g., IR False Color) confirmed the presence of ultramarine in certain passages, but the identity of other blue areas remained uncertain. Non-invasive micro-Raman spectroscopy was also considered, but the high fluorescence from the paint surface often obscured the results. In this case, definitive answers were achieved through the analysis of two micro-samples. As shown in the two cross-sections observed in UV light (Fig. 3), analysis of sample VM2 (A) revealed small particles near the surface, characterized by their typical glassy appearance. A finding confirmed by subsequent SEM-EDS and Raman analysis on the cross-section. Surprisingly, the micro-sample from the blue mantle on the Madonna’s legs (B) told a different story. Despite its blue appearance in visible light, no actual blue pigment particles were identified. The color is instead an optical effect produced by a mixture of white, black (likely vine black), and red lake particles. This case underscores a critical point: relying solely on chromatic appearance for interpreting XRF data can be profoundly misleading. It highlights a strategic analytical pathway: pXRF provided initial elemental clues but was insufficient for compound identification. Multispectral imaging suggested possibilities, while micro-sampling followed by SEM-EDS and Raman spectroscopy on cross-sections delivered the definitive, layer-specific identification of the materials. When non-invasive techniques reach their limit, targeted micro-sampling within a multi-analytical framework becomes indispensable for conclusive answers.